Make your own potato clock.
How does it work?
Each potato works as a galvanic cell, releasing electrical energy through chemical reactions. The potato juice acts as the electrolyte, in which charged atoms and molecules, called ions, dissolve and can flow over time. Wiring the potato cells end-to-end makes a series circuit, pulling the stream of electrons through the clock.
Ingredients:
- Two large, clean potatoes
- Two galvanized zinc nails
- Two copper wires
- Three jumper wires (with crocodile clips on each end)
- A battery operated
- LCD clock
STEP 1
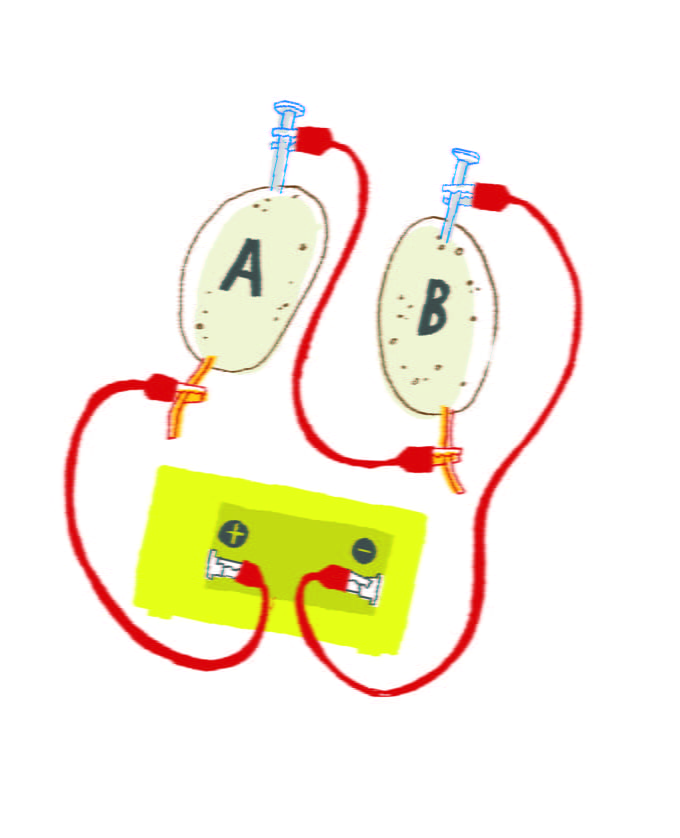
Label one potato ‘A’ and the other ‘B’.
STEP 2
Place one galvanized nail in each potato.
STEP 3
Place one copper wire piece into each potato (placing it as far away from the galvanized nail as you can).
STEP 4
Open the battery compartment of the clock and remove the battery.
STEP 5
Connect the first jumper wire from the copper wire of potato A to the positive terminal of the clock.
STEP 6
Connect the second jump wire from the galvanized nail of potato B to the negative terminal of the clock.
STEP 7
Connect the third jump wire from the galvanized nail of potato A to the copper wire of potato B.
STEP 8
Check the clock. It should now be running on potato power.