I’ve curated a stunning collection of 100 Google Earth images from the illustrious book Overview for your viewing pleasure! …
These books demystify complex concepts, illustrate the application of math in various fields, and share stories of mathematical discovery that inspire awe and wonder….
The 20+ history of science books listed below promise more than just an academic foray into the chronicles of scientific inquiry; they serve as a testament to the nobility of…
In this compilation, I present the top 10 Islamic geometry books, each offering a unique perspective on this captivating subject….
The books about bees listed here are but a few samples of the delightful troves of bee literature that await the curious reader. As you turn the pages of these…
This list of books about application of mathematics illuminates the diverse applications of mathematics, showcasing how this abstract science shapes our reality, influences various fields of research, and addresses complex…
In this guide, I delve into the best 5 graphing calculators that stand out in their field, helping engineers and students alike to achieve their academic and professional goals effectively….
Below I’ve curated a list of the seven best artificial intelligence books, perfect for those keen to learn about the intriguing world of AI, its capabilities, mysteries, and its potential…
A love for science often begins with a captivating book and the ones listed here have the power to ignite that spark. These science books are not only the highest-rated…
Help us exist with
Patreon
Get Abakcus straight to your inbox
Featured Directories

10+ Beautiful Math Tools That Make Your Math Journey Better
Mathematics is a core part of the curriculum in most schools. As such, students need to understand and appreciate the importance of math tools as they embark on their journey…

13 Official ACT Practice Tests | Full-Length & Free
For the ACT, you'll want to take at least four or five sample practice tests to get a feel for the format. Would you consider running a half-marathon if you…
Best Products
When it comes to buying any kind of tech, it’s easy to pick the wrong thing. That’s where our experts and testing team come in to help guide your buying decisions.
Watch

Toposcape
An audiovisual piece made up of over four thousand still images pulled from Google Earth, individually edited, and then manually sequenced to Midnight by Caravan Palace.

Braving the Chill: -37F Winter in Yellowstone National Park
Winter in Yellowstone transforms into a land of extremes: the collision of boiling water from the park’s famed geysers with the Frost King’s touch creates an ethereal tableau of steam…
- Nature ,

The Physics of Kung Fu
In an intersection of martial arts and digital artistry, Tobias Gremmler, a German visual artist, captures the spirit of Kung Fu through his captivating digital renderings.
Tools
Rocks
Neal Agarwal's website neal.fun/rocks, is a fantastic way to relax. With Neal Agarwal's Rocks, you are transported to a peaceful environment where you can stack rocks. This game is a…
untools
Tools for better thinking. Collection of thinking tools and frameworks to help you solve problems, make decisions and understand systems.
Featured Books
Listen

Stuff To Blow Your Mind
Stuff To Blow Your Mind is a beautiful podcast about neurological quandaries, cosmic mysteries, evolutionary marvels and our future.
- Science ,

The Numberphile
Interviews with people who love numbers and mathematics. Hosted by Brady Haran, maker of the Numberphile series on YouTube.
- Education ,
- Geometry ,
- Mathematics ,

The Skeptics’ Guide to the Universe
The world of science can be stuffy and serious, but the hosts of The Skeptics’ Guide to the Universe have found a way to make it both informative and hilarious.…
DIY
How to Construct the Spiral of Theodorus?
Constructing the spiral of Theodorus might be a good classroom activity. We have prepared the instructions for you.

How to Make LEGO Macintosh Classic with an e‑paper Display
This post is about taking things too far while playing LEGO with your kid. Ingredients: LEGO Digital Designer LEGO bricks (pick a brick) e-paper display Raspberry Pi Zero W Power…
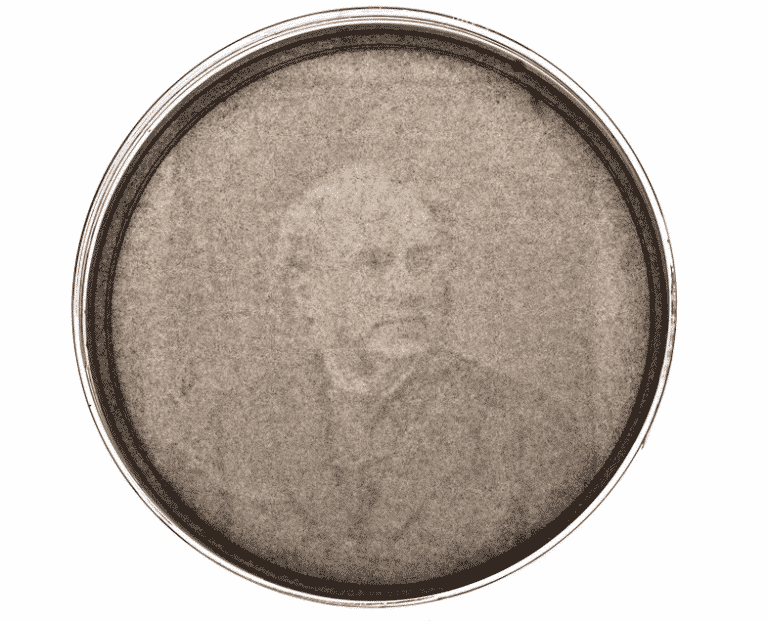
How To Grow Your Own Photo?
Bioengineer Chris Voigt have hacked a harmless strain of Escherichia coli so that it produces an organism that behaves like film.